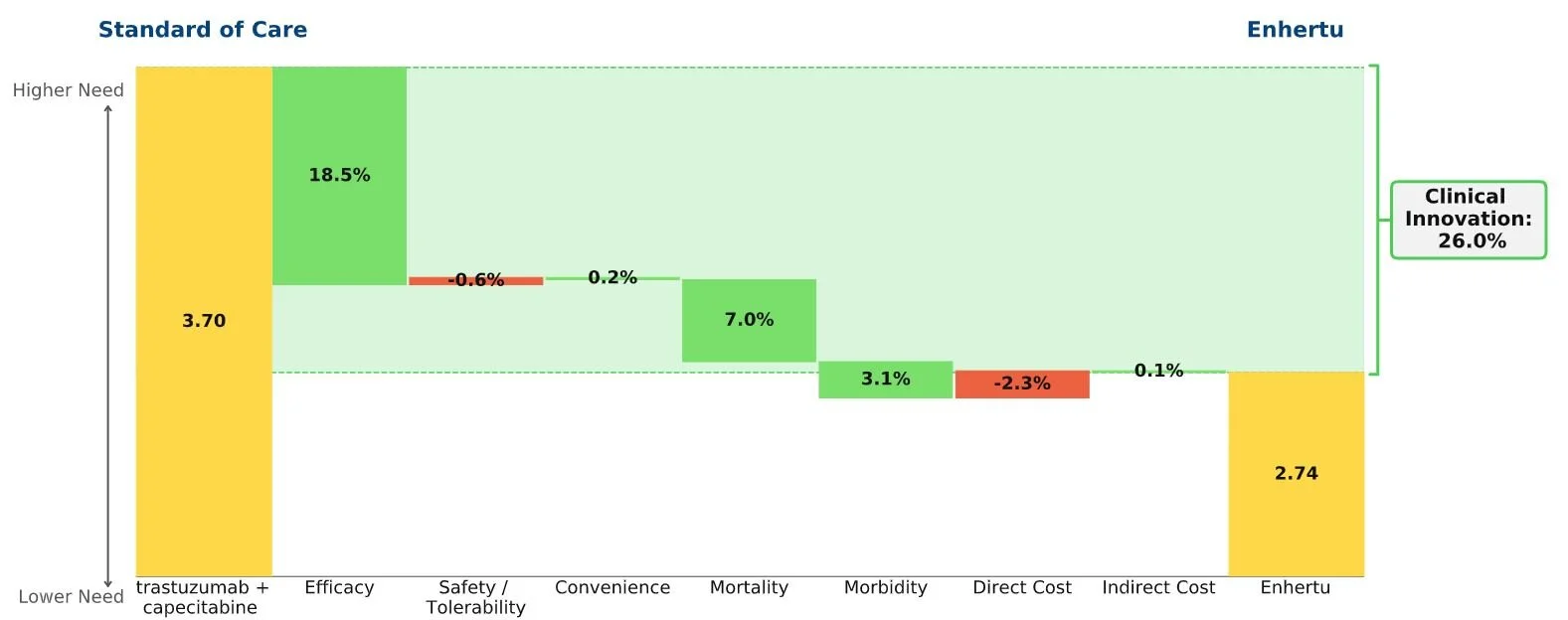
Conclusion: Enhertu achieves an impressive clinical innovation score for its substantial efficacy improvements over the current SOC (trastuzumab + capecitabine) for adults with HER2+ metastatic breast cancer who have failed multiple prior therapies.
Enhertu (trastuzumab deruxtecan, Daiichi-Sankyo and AstraZeneca) was approved in December 2019 for HER2+ unresectable or metastatic breast cancer patients who have received multiple prior anti-HER2 treatments. It boasts a significant increase in progression-free survival compared to the current standard of care (SOC), trastuzumab + capecitabine (16.4 vs. 5.6 months), as well as a notable improvement in overall response rate (60.9% vs. 22.8%). While survival data is not yet mature, even the most conservative of estimates sees Enhertu winning the day for HER2+ patients eligible for a third or later line of treatment.
The waterfall chart below shows that Enhertu’s improvements in efficacy far outweigh its higher cost and slightly worse side effect profile when compared to the current SOC. The jump in efficacy, as well as its trickle-down effect on mortality and morbidity, deliver a strong clinical innovation score of 26%.
We expect Enhertu to dominate treatment in this setting within two years. Analysts’ estimates of peak sales around $2.5 billion are reasonable.